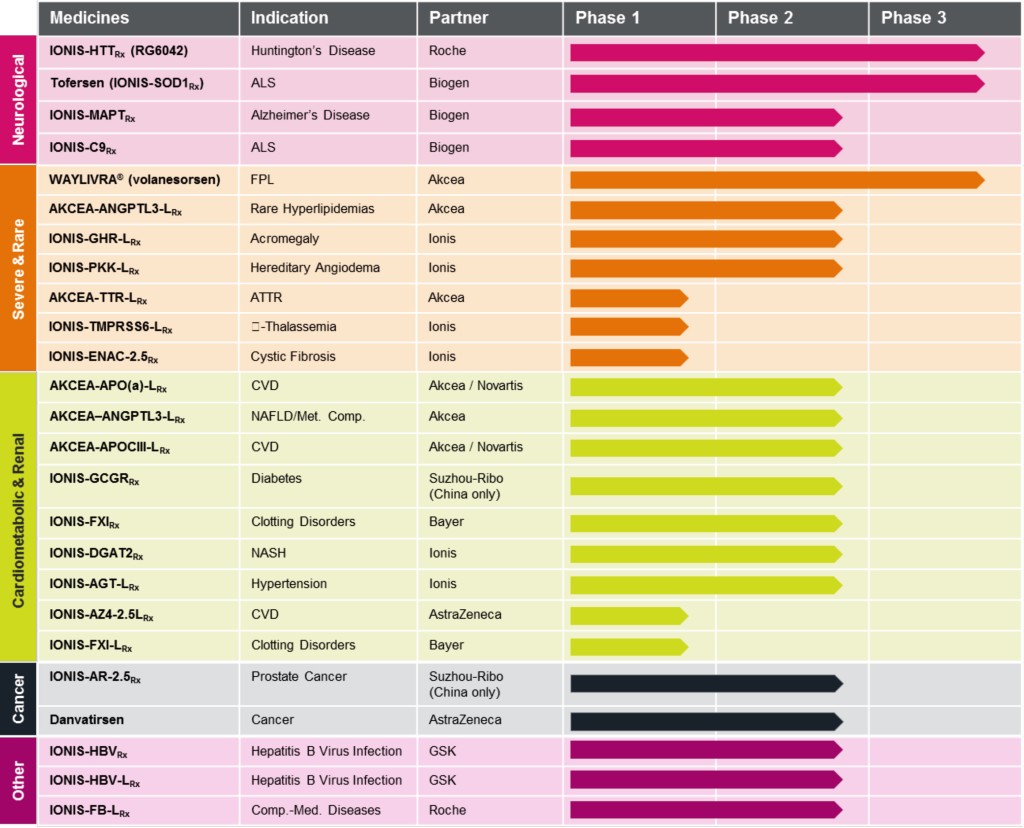
Biogen Announces Positive Phase 1 Results, Plan to License and Develop Ionis Pharmaceuticals' Investigational Therapy BIIB067 for Familial ALS - Muscular Dystrophy Association

Biogen Exercises Option with Ionis to Develop and Commercialize Investigational ASO for SMA | Biogen

Ionis on X: "Ionis initiates pivotal Phase 3 clinical study of olezarsen in patients with severe hypertriglyceridemia. Read the press release here: https://t.co/nC2SkQuKoA #antisense $IONS https://t.co/hprPaX8TEi" / X
Ionis and Akcea Enter into Strategic Collaboration with Global Pharmaceutical Company to Develop and Commercialize AKCEA-APO(a)-L Rx and AKCEA-APOCIII-L Rx | Ionis Pharmaceuticals, Inc.
IONIS-HTT Rx (RG6042) Granted PRIME Designation by the European Medicines Agency for the Treatment of People with Huntington'