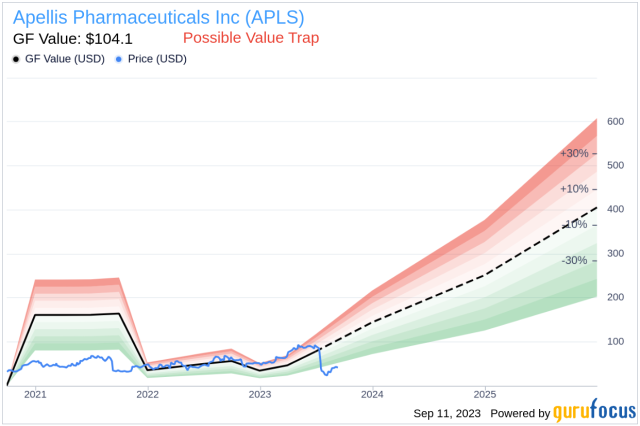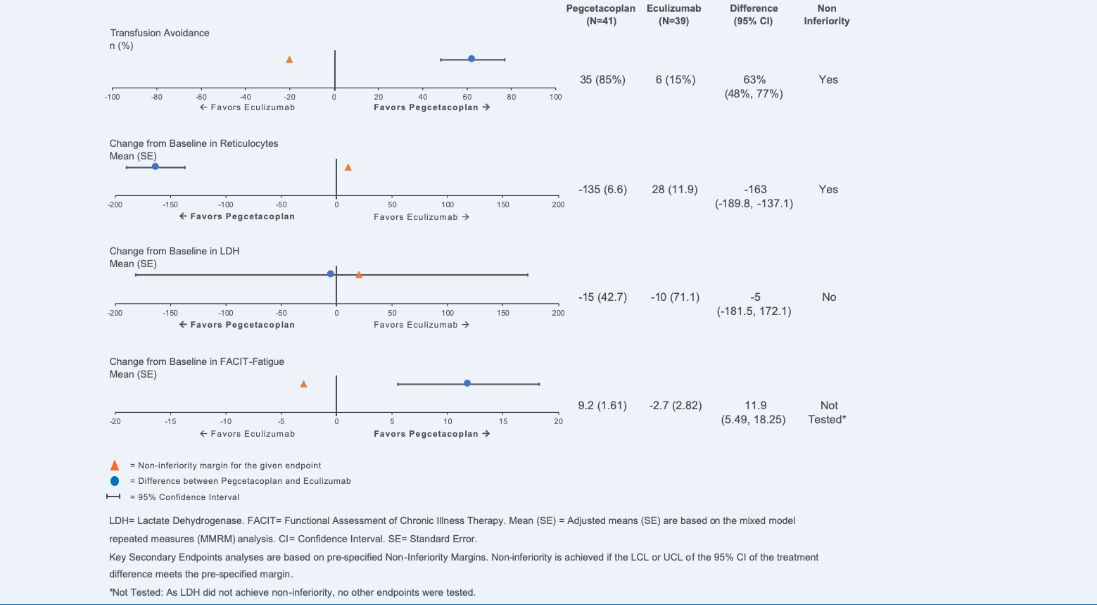
Apellis Reports Positive Top-line Results from Phase 3 Head-to-Head Study of Pegcetacoplan (APL-2) Compared to Eculizumab in Patients with Paroxysmal Nocturnal Hemoglobinuria (PNH) – Epidarex

Is There a Regulatory Strategy Lesson in the FDA's Recent pegcetacoplan (SYVFORE) approval for Geographic Atrophy : r/RegulatoryClinWriting

Apellis Provides Update on Ongoing Regulatory Review of Pegcetacoplan for GA in the European Union | APLS Stock News

Apellis Reports Results of Pegcetacoplan in P-III (DERBY and OAKS) Studies for Geographic Atrophy (GA)

APLS INVESTOR DEADLINE: Robbins Geller Rudman & Dowd LLP Announces that Apellis Pharmaceuticals, Inc. Investors with Substantial Losses Have Opportunity to Lead Class Action Lawsuit | Business Wire

Apellis Pharmaceuticals Expands Leadership Team, Appoints Adam Townsend as Chief Commercial Officer - Apellis Pharmaceuticals, Inc.

Apellis Pharmaceuticals, Inc. (APLS) Investigation: Bronstein, Gewirtz & Grossman, LLC Encourages Investors to Seek Compensation for Alleged Wrongdoings

Apellis stops dosing of pegcetacoplan in ALS trial after data review | No safety concerns, but early results don't support continuing study | ALS News Today
%20copy_1661347616.jpg)
Apellis Announces 24-Month Results in Phase 3 Studies Examining Pegcetacoplan in Geographic Atrophy (GA)

Apellis Pharmaceuticals on LinkedIn: Apellis Announces Negative CHMP Opinion for Pegcetacoplan for GA in the…
Apellis Announces U.S. FDA Approval of the EMPAVELI® Injector, a Device to Streamline Self-Administration - Apellis Pharmaceuticals, Inc.

Apellis expands R&D collaboration with Affilogic to develop targeted complement therapies for delivery into the brain - Atlanpole Biotherapies : Atlanpole Biotherapies