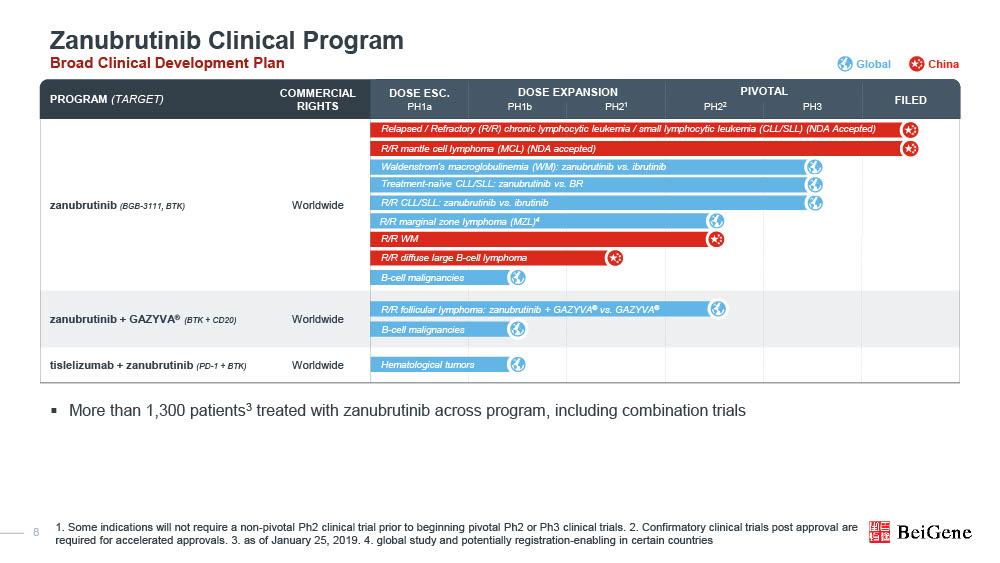
U.S. FDA Grants BRUKINSA® (Zanubrutinib) Accelerated Approval in Relapsed or Refractory Marginal Zone Lymphoma – NASDAQ (US) Website

BeiGene Announces Positive Regulatory Updates in Europe and the U.S. After Recently Regaining Global Rights for TEVIMBRA® | BGNE Stock News

BeiGene and NewBridge Pharmaceuticals Announce Approval in Saudi Arabia of BRUKINSA | NewBridge Pharmaceuticals
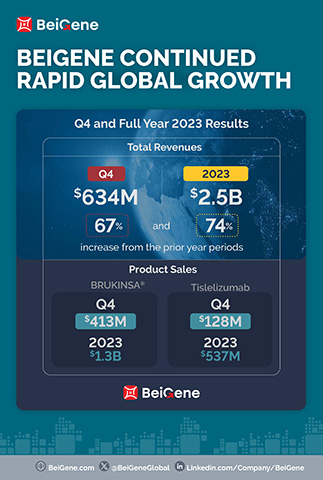
BeiGene Reports Fourth Quarter and Full Year 2023 Financial Results and Business Updates | Business Wire

BeiGene Announces First Commercial Manufacturing Approval for Its State-of-the-Art Biologics Facility in Guangzhou, China – NASDAQ (US) Website

BeiGene accueille au sein de son conseil d'administration, Olivier Brandicourt, dirigeant expérimenté dans le domaine des sciences du vivant | Business Wire