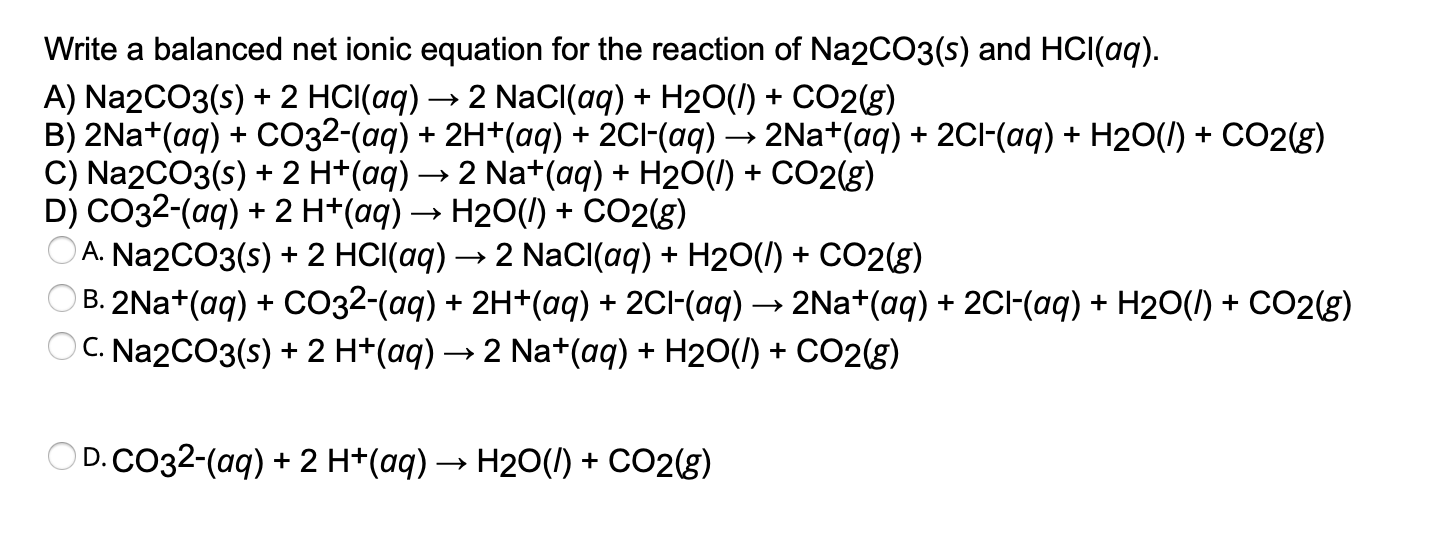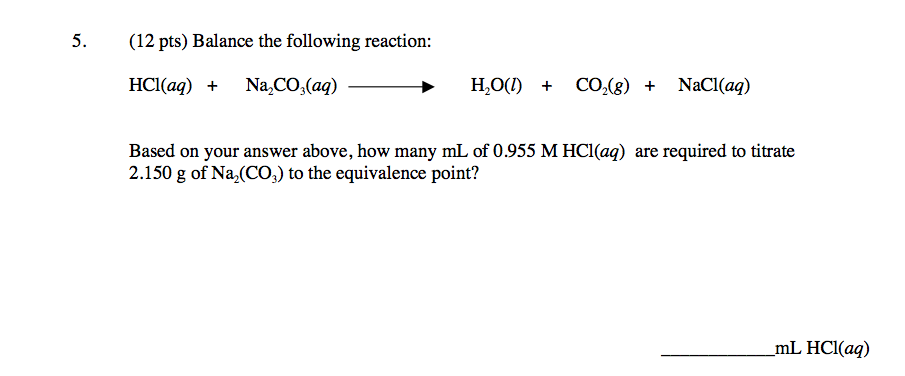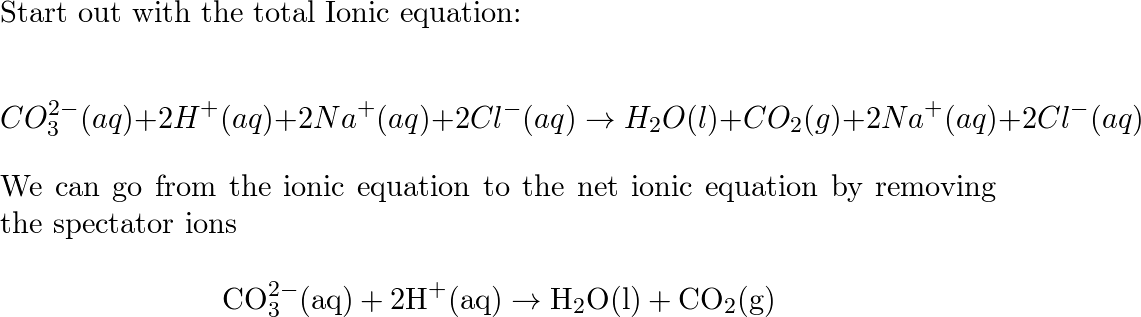Sodium Carbonate + Hydrochloric Acid - Na2CO3 + HCl - Molecular Equations & Net Ionic Equations - YouTube

write the word equation and balance the equation Na2CO3 + HCl - NaHCO3 + HCl - Science - Metals and Non-metals - 14764071 | Meritnation.com

SOLVED: Consider the following balanced chemical equation: 2 HCl(aq) + Na2CO3(aq) -> 2 NaCl(aq) + CO2(g) + H2O(l) What is the correct equilibrium equation for this reaction? Na2CO3 + 2 HCl -> 2 NaCl + CO2 + H2O

SOLVED: balance HCl+Na2CO3 and indicate physical states for all reactants and products.Then write a balanced net ionic equation for this equation including physical states

Question Video: Determining the Products Formed from the Reaction between Sodium Carbonate and Hydrochloric Acid | Nagwa

SOLVED: Hydrochloric acid (HCl) reacts with sodium carbonate (Na2CO3), forming sodium chloride (NaCl), water (H2O), and carbon dioxide (CO2). This equation is balanced as written: 2HCl(aq)+Na2CO3(aq)→2NaCl(aq)+H2O(l)+CO2(g) Part A What volume of 1.75

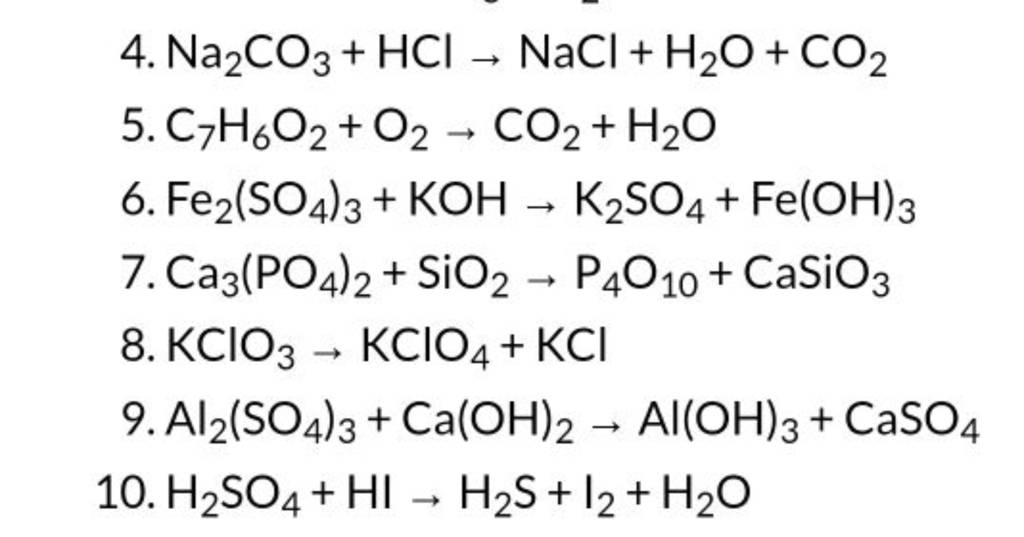
SOLVED: The balanced chemical equation for the reaction occurring is: 2 HCl + Na2CO3(aq) -> 2 NaCl + H2O + CO2(g) What is the mole ratio for hydrochloric acid and sodium carbonate?