PLX - Protalix BioTherapeutics, Inc. Stock - Stock Price, Institutional Ownership, Shareholders (NYSEAM)
Protalix Biotherapeutics Stock News | PLX Company Stock News and Press Releases | FinancialContent Business Page

Protalix Biotherapeutics and Chiesi Global Rare Diseases Provide Regulatory Update on PRX-102 for the Treatment of Fabry Disease

Protalix BioTherapeutics and Chiesi Farmaceutici Announce Receipt of "Agreement Letter" for Initial Pediatric Study Plan for PRX-102 for the Treatment of Fabry Disease
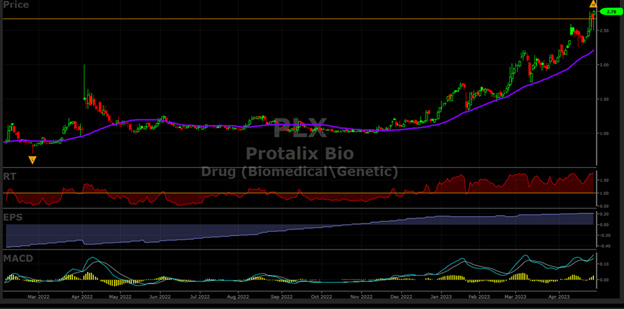
Will Protalix BioTherapeutics Keep Powering Higher After its 178% Run Up Since November 2022? - VectorVest

Protalix BioTherapeutics Expands Partnership with Chiesi Farmaceutici to Include Exclusive US Rights for the Development and Commercialization of PRX-102 (pegunigalsidase alfa)